|
The weaker electrostatic attraction of the electron being gained to the positive nucleus, makes it harder for the atom to gain an electron and it decreases in reactivity in group 7. Therefore, the attraction of the outer electron to the nucleus decreases as you go down group 7. There are more electrons shielding the positively charged nucleus from the outer electron. They increase in electron shells so the atoms are larger as you go down the group. The atomic mass of the halogens increases.As you go down group 7, reactivity of the halogens decreases because: The negatively charged electron is attracted to the halogen atom due to the positive charge of the atom’s nucleus. Halogens react to gain an electron so they become more stable. Group 7: Reactivity (GCSE Chemistry) Group 7: Reactivity Halogens – trend in reactivityĪs you look at the trend in reactivity of group 7, the reactivity of halogens decreases so fluorine is the most reactive halogen and astatine is the least reactive halogen. 
Chemistry Teacher Resource Edexcel | AQA | OCR.Biology Teacher Resource Edexcel | AQA | OCR | CIE.Booking A-Levels as a Private Candidate.Chemistry Flashcards Edexcel | AQA | OCR | CIE.Biology Flashcards Edexcel | AQA | OCR | CIE.Chemistry Textbook Edexcel | AQA | OCR | CIE.Biology Textbook Edexcel | AQA | OCR | CIE.Physics Weekly Classes Edexcel | AQA | OCR | CIE. 
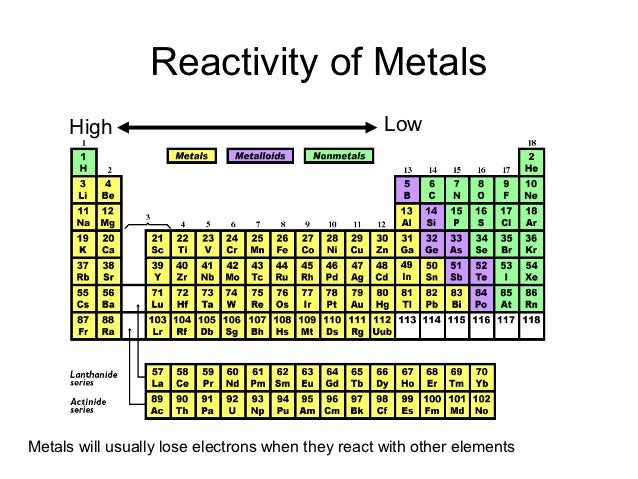
In the below periodic table you can see the trend of.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
